Good laboratory practices (GLP) and good manufacturing practices (GMP) are two sets of standards that play an essential role in consumer product quality control and assurance. While both are related to the manufacturing process, they have distinct differences.
This article will explore the differences between GLP and GMP, their roles in product safety, and the implications of not following the respective standards.

Additionally, this article will look at the history of GLP and GMP, and how they have evolved over time. We shall also learn, how they can be implemented in a modern manufacturing setting. By the end, readers will have a better understanding of the importance of GLP and GMP and the impact they have on product quality and safety.
Additionally, ERP.AI can support compliance by helping manufacturers automate documentation, monitor quality metrics in real-time, and ensure every step of production aligns with both GLP and GMP standards.
Overview of GMP and GLP
Good Manufacturing Practice (GMP) is a system of processes and procedures that pharmaceutical and medical device manufacturers must adhere to in order to ensure that their products are of the highest quality. This includes the production, quality control, packaging, and storage of products. GMP regulations are designed to ensure that products are safe, effective, and high-quality.
Good Laboratory Practice (GLP) is a set of principles and guidelines for laboratory operations. It is designed to ensure that the results of laboratory experiments and studies are reliable, reproducible, and of high quality. GLP regulations cover the people, premises, equipment, materials, and processes used in a laboratory. They also include requirements for the documentation and reporting of experiments and studies.
How do GMP and GLP Weigh in Pharmaceutical Manufacturing?
GMP and GLP are quality standards applied to the manufacturing of pharmaceuticals and other healthcare products. GMP covers the manufacturing process and the quality of the products while GLP covers the laboratory testing of samples, data management, and other aspects of research and development. GMP and GLP are important for ensuring the safety and efficacy of pharmaceutical products.
Both these practices are two of the most important regulations in pharmaceutical manufacturing. GMP refers to a set of rules and regulations enforced by the US Food and Drug Administration (FDA) that cover the entire drug manufacturing process. It takes care of procedures from raw material acquisition and handling to the finished product.

It includes quality assurance, quality control, process control, and personnel qualification requirements. GMP also emphasizes the necessity of regularly monitored and updated standard operating procedures (SOPs).
The Organization for Economic Cooperation and Development created GLP regulations (OECD). It specifies the requirements for non-clinical safety studies' design, operation, and monitoring.
GLP encompasses the entire drug development process, including experiment design, the use of laboratory animals, data collection and storage, and results reporting. GLP is intended to ensure that the data generated by experiments is of high quality and reproducible.
GMP and GLP are both required to ensure the quality and safety of pharmaceutical products. GMP ensures that drugs are manufactured safely and consistently, whereas GLP ensures that the data collected during the drug development process is of the highest grade. They are both necessary for ensuring drug safety and effectiveness.
Key Differences between GMP and GLP
While the two systems have many similarities, there are also some key differences that set them apart. In this section, we will discuss the main differences between GMP and GLP and how they can affect the overall production and quality of products.
Purpose
GMP is a system of guidelines developed to ensure that drug products are of quality. On the other hand, GLP is a system of regulations designed to ensure that data generated from non-clinical laboratory and animal studies are of quality.
Nature
GMP is related to the manufacturing, quality control, and quality assurance of drugs. Whereas, GLP is related to the control of laboratory practices for non-clinical animal studies.
Enforceability
GMP is enforceable by both the FDA and the European Medicines Agency, while GLP is only enforceable by the FDA.
Documentation
GMP has documentation requirements relating to manufacturing, quality control, and quality assurance. On the other hand, GLP has documentation requirements relating to laboratory practices and the reporting of non-clinical studies.
Regulatory Requirements
GMP has regulatory requirements regarding personnel qualifications, facility requirements, production and process controls, and quality assurance. Whereas, GLP has regulatory requirements regarding laboratory facility requirements, laboratory practices, and reporting.
GMP Regulation
This section will focus on the history and requirements of GMP regulation.
A. History of GMP
The history of GMP Regulation dates back to the late 19th century. In the United States, the Pure Food and Drug Act was passed in 1906, which tasked the Bureau of Chemistry to enforce standards of purity and safety for food and drug products.
This was the first attempt to regulate the production and distribution of food and drugs. In the 1950s, the US Food and Drug Administration (FDA) began to establish Good Manufacturing Practices (GMPs), which set minimum standards for the production of pharmaceuticals and medical devices.
These GMPs included requirements for manufacturing processes, quality control, and document control. In the 1960s, the FDA issued revised GMP regulations and began to enforce them.

In the 1970s, the FDA issued a new set of GMP regulations, which included additional requirements for production, testing, and record-keeping. In the 1980s, the FDA revised its GMP regulations to include stricter requirements for the production and distribution of pharmaceuticals and medical devices. This included requirements for clean rooms, air quality, and employee training.
In the 1990s, the FDA began to focus on the safety of food and drugs and issued new GMP regulations that included more stringent requirements for product testing and documentation.
B. Requirements of GMP
GMP are regulations set forth by the FDA and other regulatory agencies to ensure that medical devices and other products comply with safety standards.
These regulations are designed to help protect public health by ensuring that products are correctly manufactured, labeled, and stored. GMP regulations require manufacturers to follow specific practices for product development and production, QC, personnel training, and documentation.
They also require that manufacturers regularly inspect and test their products to confirm that they meet safety, effectiveness, and quality standards. Additionally, GMP regulations require manufacturers to keep detailed records of their operations and submit them for FDA review.
C. Examples of GMP
Let’s look at some areas where GMP is applied:
- Establishing and following written procedures for production and process control
- Document all activities relating to the production and control of products
- Ensuring accurate and complete records of all production activities
- Maintaining clean and hygienic premises and equipment
- Ensuring that all personnel involved in manufacturing, testing, and releasing products are qualified and trained
- Ensuring that all products are correctly identified and traceable throughout the manufacturing process
- Establishing and following appropriate sampling and testing procedures
- Ensuring that all raw materials and finished products meet the specified requirements
- Implementing and monitoring systems for product recall, customer complaints, and product quality reviews
- Establish and follow procedures for handling, storing, and distributing products
GLP Regulation
Having learned about the GMP, let’s glide through the GLP regulation and its diverse aspects.
A. History of GLP
GLPs originated in the United States in the 1970s as a response to the lack of consistency and reliability of some studies conducted in the pharmaceutical industry. GLPs ensure that laboratory studies are conducted in a way that minimizes the risk of bias and error, and that the results are reproducible and trustworthy.
GLPs set out requirements for the organization and management of a laboratory, the qualifications and training of personnel, the quality assurance of equipment and materials, the design and conduct of studies, and the reporting and storage of data.
GLPs are now internationally recognized and are used by regulatory authorities around the world. These practices are implemented to evaluate the safety and efficacy of drugs and other products.
B. Requirements of GLP
In the previous section, we understood that GLP is designed to ensure that laboratory studies are conducted and reported accurately and reliably. It requires establishing and maintaining documented standard operating procedures (SOPs) which specify the responsibilities of personnel involved in the study, and the proper handling of materials and data.
It also requires companies to ensure that personnel is qualified and trained in the techniques used in the study. Organizations must assign personnel to specific tasks and responsibilities. They must take care that test facilities are suitable and safe for use in the study.
They must maintain records of all raw data, calculations, and observations. This way, they can ensure that all data is traceable and accessible.
Teams must also check that all data is reviewed and approved prior to its release. The requirements also include ensuring that the study is conducted in a manner that ensures the integrity of the data and results.
C. Examples of GLP
The following points enumerate the examples of GLP:
- Training personnel in standard operating procedures (SOPs) and following these procedures
- Establishing and following a system for maintenance of records and maintaining records for a minimum of 10 years
- Establishing and following a system for the calibration and maintenance of lab equipment
- Documenting all changes to the experimental protocol and obtaining approval before proceeding with the experiment
- Ensuring that all data is accurate and not subject to subjective interpretation
- Investigating all discrepancies in data and documenting the results
- Ensuring that all materials used in the experiment are of the highest quality and properly stored
- Documenting all deviations from the experimental protocol and obtaining approval before proceeding with the experiment
- Performing all experiments according to established protocols and in a timely manner
- Conducting regular reviews of all SOPs, protocols, and data.
Benefits of GMP and GLP
Now that we have gathered the requirements of GMP and GLP, let’s also learn about the benefits of these practices.
Benefits of GMP
- Ensures that products are consistently produced and controlled according to quality standards
- Helps to prevent contamination, mix-ups, and other errors
- Minimizes risks to consumers, as well as to the environment
- Improves product quality and safety
- Increases efficiency of production management and results in cost-effective production
- Creates an environment where employees can work safely and securely
- Enhances customer satisfaction
Benefits of GLP
- Ensures that results are reliable and accurate
- Minimizes the risk of contamination or errors
- Enhances the credibility of research data
- Helps to ensure compliance with regulations
- Ensures that laboratory personnel is properly trained
- Improves the efficiency of laboratory operations
- Enhances safety in the laboratory environment
Quality Assurance with GMP and GLP
GMP and GLP are essential to any quality assurance program. GMP ensures that products are manufactured consistently and safely, and that quality is consistently maintained throughout the production cycle.
GLP ensures that laboratory studies are conducted in a consistent, reliable, and reproducible manner. Together, GMP and GLP provide the foundation for a quality assurance program by ensuring that products are produced and tested in a safe, consistent, and reliable manner.
Cost Savings with GMP and GLP
GMP and GLP can help to reduce costs in a number of ways. Firstly, they provide a framework of procedures and controls that help to ensure the quality and safety of products and processes. This helps to prevent costly errors and recalls that can occur if the quality is not properly maintained.
Additionally, GMP and GLP help to ensure that production planning is efficient and streamlined, leading to reduced waste and improved productivity. Finally, these practices provide a standardized approach to production that allows for better cost control and the ability to identify and reduce unnecessary expenses.
Better Efficiency with GMP and GLP
By following GMP and GLP guidelines, businesses can improve efficiency by reducing waste, improving accuracy, and ensuring compliance with applicable laws and regulations. Additionally, GMP and GLP guidelines can help businesses save money by reducing the cost of raw materials, minimizing product recalls, and eliminating the need for costly rework.
When to Use GMPs and GLPs?
GMPs are used to ensure that a product is safe, effective, and of high quality. GLPs are guidelines set forth by the FDA that are used to ensure the reliability and accuracy of data generated in a laboratory setting.
GLPs are used to ensure that data is reproducible and that experiments are conducted and documented in a consistent manner. GMPs are used in the manufacturing process of pharmaceuticals and other products, while GLPs are used in laboratory settings such as research and development, quality control, and safety evaluations.
GMPs focus on the quality of the product, while GLPs focus on the reliability of the data.
Implications of not following GMP and GLP
GMP and GLP) are standards to ensure the safety and quality of products created in the pharmaceutical, biotech, and medical device industries. Failure to follow GMP and GLP can have a variety of implications, including the following:
- Loss of Trust: Loss of customer confidence and trust in the products produced by the company, leading to decreased sales and potential loss of business.
- Risk of Contamination: Undetected or unidentified product defects or contamination that could result in serious harm or injury to consumers.
- Fines and Penalties: Fines and/or legal action from regulatory agencies for non-compliance.
- Damage to Reputation: Reputation and brand damage from negative publicity related to the company’s failure to adhere to GMP and GLP.
- Losses: Financial losses due to product recalls and other corrective actions.
How AI Improves Manufacturing Systems
From automating routine tasks to enabling predictive analytics, AI enhances every stage of the production cycle. It analyzes real-time data to forecast demand, optimize inventory levels, and reduce machine downtime through predictive maintenance. This leads to smoother operations and fewer disruptions.
AI plays a key role in this transformation by integrating AI with manufacturing workflows—allowing businesses to gain deep insights into their supply chain, automate production planning, and fine-tune resource allocation.
GMP and GLP FAQs
There are some questions that are frequently asked about GMP and GLP. Let’s check them out here:
Q: What are GMPs and GLPs?
A: GMPs (Good Manufacturing Practices) and GLPs (Good Laboratory Practices) are sets of standards and regulations for the manufacturing, development, and testing of products. They are designed to ensure that products are safe, effective, and of high quality.
Q: What are the differences between GMPs and GLPs?
A: GMPs are designed to ensure the safety and quality of manufactured products, while GLPs are designed to ensure the accuracy and reliability of laboratory experiments.
Q: Who enforces GMPs and GLPs?
A: GMPs and GLPs are enforced by regulatory agencies such as the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Q: What are the consequences of not complying with GMPs and GLPs?
A: Not complying with GMPs and GLPs can result in product recalls, fines, and other penalties.
How can Deskera Help You?
The pharmaceutical and healthcare sectors are looking for a comprehensive system that will allow them to align all of their business modules, including accounting, inventory, and employee management.
With Deskera, you can benefit from a distribution-focused ERP system that is future-proof for a wide range of businesses. The best part is that it can be tailored to your specific needs. Our ERP gives you a forward-thinking perspective on how to grow and advance your business.
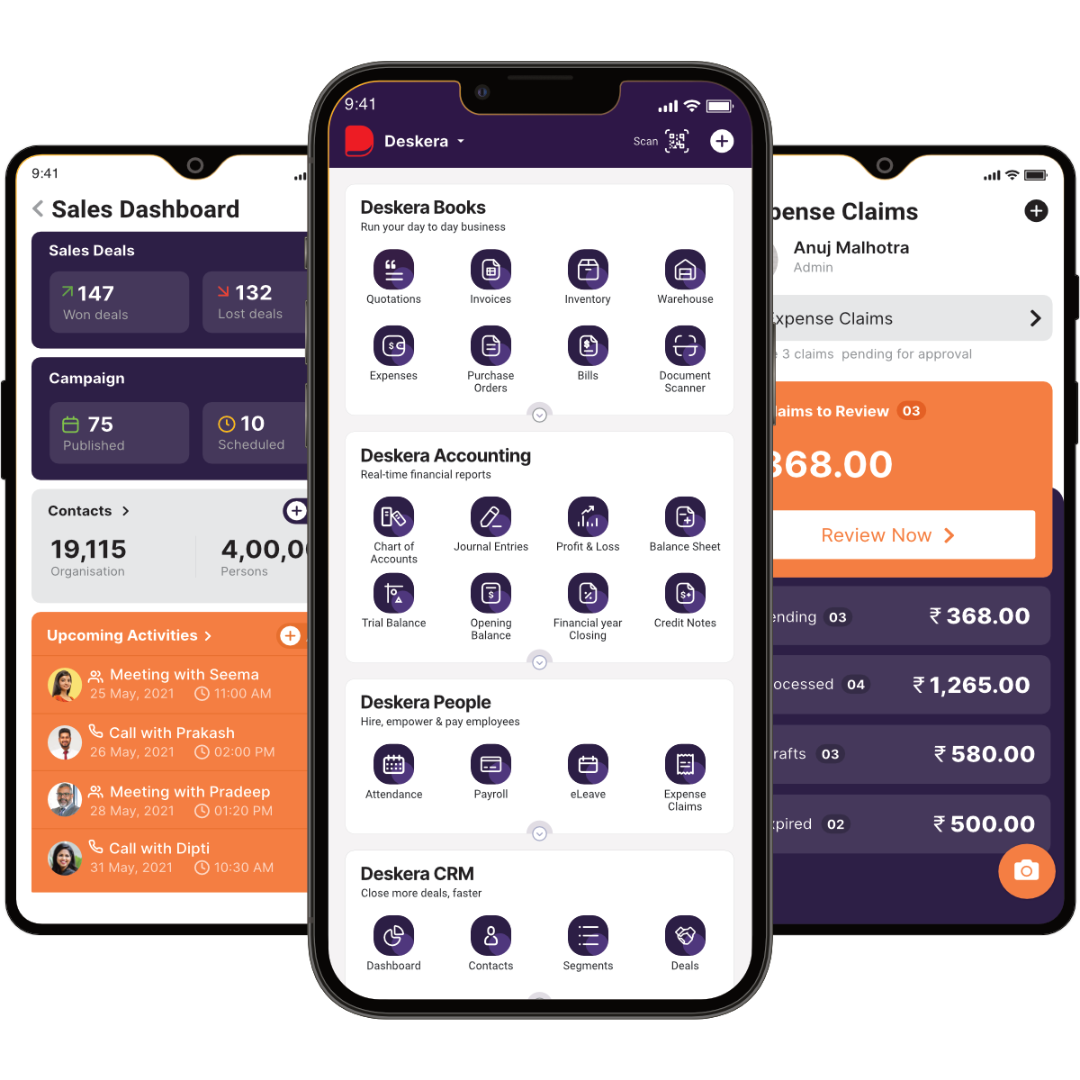
Deskera Books allows you to automate all major accounting tasks such as invoicing, expenses, and billing.
Deskera People is a specialized tool for managing employee payroll, deductions, attendance, and leaves.
Deskera CRM includes all of the solutions that can help you grow your customer base, such as CTAs, landing pages, email marketing, and so on.
Key Takeaways
- Good laboratory practices (GLP) and good manufacturing practices (GMP) are two sets of standards that play an essential role in consumer product quality control and assurance.
- Good Manufacturing Practice (GMP) is a system of processes and procedures that pharmaceutical and medical device manufacturers must adhere to in order to ensure that their products are of the highest quality.
- Good Laboratory Practice (GLP) is a set of principles and guidelines for laboratory operations. It is designed to ensure that the results of laboratory experiments and studies are reliable, reproducible, and of high quality.
- GMP and GLP are quality standards applied to the manufacturing of pharmaceuticals and other healthcare products.
- GMP covers the manufacturing process and the quality of the products while GLP covers the laboratory testing of samples, data management, and other aspects of research and development.
- GMP is a system of guidelines developed to ensure that drug products are of quality. On the other hand, GLP is a system of regulations designed to ensure that data generated from non-clinical laboratory and animal studies are of quality.
- GMP is related to the manufacturing, quality control, and quality assurance of drugs. Whereas, GLP is related to the control of laboratory practices for non-clinical animal studies.
- GMP is enforceable by both the FDA and the European Medicines Agency, while GLP is only enforceable by the FDA.
- GMP has documentation requirements relating to manufacturing, quality control, and quality assurance. On the other hand, GLP has documentation requirements relating to laboratory practices and the reporting of non-clinical animal studies.
- Not abiding by the GMP and GLP can lead to a loss of trust from customers and attract fines and penalties.
- Also, there may be a risk of contamination which would lead to a damaged reputation for the brand and the company.
- GMP and GLP both have their own benefits and drawbacks. GMP is cheaper and easier to implement, but it does not provide the same level of assurance as GLP. GLP can be more expensive and difficult to implement, but it provides more assurance that the product meets the regulatory requirements.
- Ultimately, the choice between GMP and GLP depends on the specific needs of a company and its products. In some cases, one may be more suitable than the other, while in others, a combination of the two may be necessary. Regardless of choice, it is important to keep in mind that the goal is to ensure that the end product meets the regulatory requirements.
- In conclusion, GMP and GLP are two different quality control standards that are used in the production of products. Ultimately, the choice between the two depends on the specific needs of a product or company and the assurance that is needed.
Related Articles